Inari Medical: A commercial stage medical device company focused on venous diseases
Profitable hypergrowth medical device company.
Blood clot appears to be a major cause for both morbidity and mortality to humans all over the world including the United States.
The major causes of death and morbidity are heart attack, stroke and pulmonary embolism (venous thromboembolism).
All these diseases are caused by blood clots. Blood clots in arteries of the brain leads to stroke and blood clots in arteries of heart leads to heart attack.
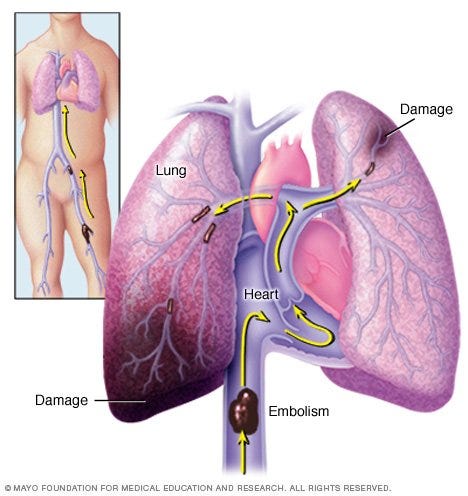
Another form of clots in veins can ultimately lead to blood clots in lungs leading pulmonary embolism which is also the cause of sudden death in humans. This is collectively called venous thromboembolism (VTE).
With this concept in the background, let's look at the current magnitude of the problem in the US.
Magnitude of VTE
Venous thromboembolism can be classified into two distinct diagnoses: blood clots in veins of legs = Deep venous thrombosis (DVT) and blood clots in lungs = Pulmonary embolism (PE).
There are 1 million people suffering from blood clots in the venous system each year. Out of 1 million, 668,000 are DVT and 400, 000 are diagnosed with PE each year.
30% of patients diagnosed with venous thromboembolism die each year!
Current standard of care of VTE
Currently health care providers utilize clot busters and or anticoagulants like heparin to treat venous thromboembolism. Anticoagulants are intended to stop further clot formation while the body is trying to absorb already formed clots.
Company products
ClotTriever: FDA cleared for the removal of clot from peripheral blood vessels and is used to treat patients suffering from deep venous thrombosis or DVT.
FlowTriever: FDA cleared for removal of blood clots from lungs.
Role of ClotTriever and FlowTriever the context of present therapy:
Historically, physical removal of blood clots from venous systems have focused on using devices designed for arteries. Due to difference in structure of arteries and veins as well as formation of different type blood clots in veins versus arteries: arterial devices have difficulty removing blood clots from veins.
Presently available clot busters are associated with risk of major bleeding in the brain, expensive, require ICU stay and are not that effective.
Market size and opportunity
About 668,000 patients in the US are diagnosed with DVT each year. About 38% of these DVTs are located in bigger veins of legs called Iliofemoral region and are candidates for treatment using ClotTreiever product. This represents around 1.6 billion opportunities.
About 400, 000 patients suffer from clots in the lungs each year. About 50% of these patients are candidates for treatment using FlowTriever. This represents 2 billion opportunities.
Collectively the company estimates a total addressable market of 3.6 billion.
This may take some investors back and easily dismiss the company as overvalued as the current market cap of the company is around 5.4 billion, currently trading at 22 times revenue of 2021 of 240 million.
Opportunity:
Although the total addressable market doesn’t appear large with US TAM of 3.6 B; there is opportunity for international expansion. DVT and PE doesn’t only affect the US population; it affects people from all over the world. The device company like NARI could have a monopoly as its products are only available devices to remove clots from the venous system. I wouldn't be surprised if they will command 50% market share in the US which will give them a revenue run rate of about 1.6 billion. The company boasts a gross margin of > 90% and could easily command 15 times revenue even at that revenue rate as international expansion will still be underway by that time. I contend that this company should command a higher revenue multiple because of high gross margin, low capex and high ROIC. This translates into prodigious free cash flow at scale. Since DVT and PE occur every year; the company’s revenue stream will be recurring. A few interventionists who have actually used the devices have DM on twitter to express their satisfaction with the devices which bodes well for the company and the investors as physicians have ultimate say regarding the selection of devices for their patients. This is always guided by the best outcomes for the patients.
I have to admit that I bought this company in July 2020 and made a mistake by selling it too early. During last month's selloff I bought shares back and I am now long.
I intentionally wanted this write up to be concise and I don’t provide in depth financial analysis as this is available in company’s website and various other websites. Further financial analyses are very subjective and sometimes leave room for disagreements. This write up’s mainly goal is to excite interest in the company. I hope each one of you do your own due diligence and decide for yourself if it is good for investment. I hope after reading this write up, you will have understanding about venous blood clot disorder and the problem this company is trying to solve.
Disclaimer: The stocks mentioned in my newsletters are not intended to be a list of buy recommendations but rather some ideas for your watchlist. Perhaps they end up in your own portfolio after you conduct your own research and due diligence. Some of the stocks mentioned in my newsletters have smaller market capitalizations and therefore can be more volatile. I always encourage everyone to do their own research and due diligence before buying any stocks mentioned in my newsletters. Please manage your portfolio and position sizing in accordance with your own risk tolerance and investment objectives.








Very nice analysis! Have they demonstrated in their studies that the retrieval of clot is superior to the use of blood thinners alone? Will the patients still need to be on blood thinners after this procedure? Lastly, how is Inari planning to overcome the barrier of providers’ hesitancy to try something new?